Medical CNC Machining Services
XC Machining produces surgical instruments, orthopedic implant components, dental abutments, and diagnostic device housings in titanium Grade 5, stainless steel 316L, cobalt-chrome, PEEK, and UHMWPE with tolerances to ±0.0127 mm and surface finishes from Ra 0.2 μm.
- All uploads are secure and confidential

What Medical Components Does XC Machining CNC Produce?
XC Machining CNC produces medical components in biocompatible titanium, surgical-grade stainless steel 316L, cobalt-chrome, PEEK, Delrin, and UHMWPE. All medical projects follow ISO 9001:2015 quality procedures with full material traceability and batch inspection documentation.
Core medical CNC applications at XC Machining include the following part categories:
- Surgical forceps, retractors, guides, and scalpel handles
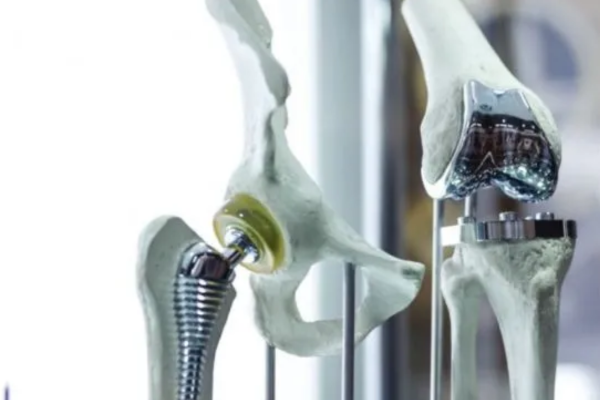
- Hip, knee, and spinal implant interface components
- Orthopedic bone plates, screws, and fixation hardware
- Dental abutments, crowns, bridges, and surgical guides
- Prosthetic joint sockets, liners, and attachment mechanisms
- Diagnostic housings, manifolds, and fluid-path connectors
What Are the Advantages of Medical CNC Machining?
Medical CNC machining at XC Machining delivers four advantages critical to medical device OEMs, surgical instrument manufacturers, and orthopedic implant developers: fast prototyping turnaround, batch-to-batch dimensional repeatability, full-service manufacturing capability under one quality system, and complex geometry production in difficult-to-machine biocompatible alloys.

Medical CNC prototypes ship in 3–5 business days for design verification testing (DVT) samples. Production batches of 100–5,000 parts complete in 10–15 business days. XC Machining schedules medical projects around regulatory submission deadlines, clinical trial timelines, and 510(k) testing cycles to prevent delays.
XC Machining holds ±0.005 mm part-to-part dimensional consistency across production runs of 100 to 10,000 medical parts. All CNC machines follow documented calibration schedules traceable to NIST standards. Every production batch receives CMM inspection with Cpk ≥ 1.33 process capability verification.

XC Machining provides 5-axis CNC milling, Swiss CNC turning, wire EDM, CNC lathe machining, medical injection molding, and metal 3D printing under one facility. Post-machining processes include passivation per ASTM A967, electropolishing, bead blasting, and laser marking for UDI compliance.
Five-axis simultaneous CNC machining produces contoured implant surfaces, anatomically shaped bone plate profiles, undercut screw pockets, and multi-angle drill guide channels in a single setup — eliminating re-fixturing error and maintaining ±0.0127 mm accuracy on all patient-contact features.
What Tolerances Does Medical CNC Machining Achieve at XC Machining?
XC Machining holds medical-grade CNC tolerances per the specifications below. Medical parts require tighter tolerances than standard CNC work — our equipment and CMM inspection systems support ±0.0127 mm precision on critical dimensions.
| Description | Specification |
| Metals | ISO 2768-m |
| Plastics | ISO 2768-c |
| Precision Tolerances | Medical CNC machining can achieve tolerances as tight as +/- 0.0005 inches (0.0127mm) or as required by your specifications. |
| Surface Finish | Ra 32 µin (0.8 µm) |
| Minimum Wall Thickness | 0.3mm |
| Minimum End Mill Size | 0.2mm |
| Minimum Drill Size | 0.6mm |
| Maximum Part Size | CNC Milling: 2000×1000×500 mm |
| CNC Turning: 150×300 mm | |
| Minimum Part Size | CNC Milling: 5×5×5 mm |
| CNC Turning: 1×1 mm | |
| Production Volume | Prototyping: 1-100 pcs |

What CNC Machining Technologies Are Used for Medical Parts?
XC Machining uses four specialized CNC technologies for medical part production. Each technology addresses specific part geometries, material challenges, and tolerance requirements in medical device manufacturing.
- Wire EDM Medical Machining
- 5-Axis CNC Medical Machining ORIGINAL
- Swiss CNC Medical Machining
- Medical CNC Milling
How Does XC Machining Customize CNC Parts for Medical Devices?
XC Machining CNC machines custom medical device components per your CAD geometry, material specification, and functional requirements. Our engineers review each design for minimum wall thickness (0.3 mm), biocompatible material selection (titanium, 316L, CoCr, PEEK), sterilization compatibility (autoclave, EtO, gamma radiation), and surface finish requirements (Ra 0.2–0.8 μm for patient-contact surfaces).

How Fast Can XC Machining Produce Medical CNC Prototypes?
XC Machining delivers medical CNC prototypes in 3–5 business days for DVT (design verification testing) samples. We produce 1 to 50 functional prototypes in production-grade titanium, PEEK, or 316L stainless steel — matching final part material properties for fit, form, and function validation before 510(k) or regulatory submission.
What Quality and Equipment Does XC Machining Provide for Medical CNC?
Every medical CNC project at XC Machining follows ISO 9001:2015 procedures with in-process CMM verification, surface roughness measurement per Ra specification, and 100% visual inspection under magnification. Medical shipments include dimensional reports, material certifications with heat lot traceability, and certificates of conformance (CoC).


Our medical machining department operates 5-axis simultaneous CNC mills, Swiss-type CNC lathes (0.5–32 mm diameter capacity), wire EDM machines, and CNC turning centers with live tooling — all on documented calibration schedules traceable to NIST measurement standards.

Medical-grade materials: titanium Grade 2 and Grade 5 (Ti-6Al-4V per ASTM F136), stainless steel 316L, 17-4 PH, cobalt-chrome (CoCr), PEEK, UHMWPE, Delrin, and medical-grade polycarbonate. Material certificates and mill test reports provided with every order.

Our engineers review medical designs for wall thickness (minimum 0.3 mm), tool access on internal features, sterilization compatibility (autoclave 134°C, EtO, gamma), surface finish targets (Ra 0.2–0.8 μm), and biocompatible material alternatives. DFM feedback within 12 hours.

Medical finishes include passivation per ASTM A967 (stainless steel), electropolishing to Ra 0.1–0.4 μm, bead blasting, mirror polishing to Ra 0.05 μm, and laser marking for UDI (Unique Device Identification) regulatory compliance.
What Surface Finishes Are Available for Medical CNC Parts?
XC Machining applies the following surface finishes to medical CNC parts for biocompatibility, corrosion resistance, sterilization compatibility, and patient-contact surface compliance.

Sanding removes CNC tool marks from medical parts to Ra 0.8–1.6 μm, preparing surfaces for electropolishing or passivation on implant and instrument components before final sterilization.

Brushing produces a directional satin texture at Ra 0.4–0.8 μm on stainless steel surgical instruments, reducing reflective glare under operating room lighting and providing improved wet-grip performance during procedures.

Electropolishing removes 10–25 μm from stainless steel and titanium medical surfaces via electrolytic dissolution, achieving Ra 0.1–0.4 μm per ASTM B912 with enhanced corrosion resistance for implant-contact applications.

Type III hard-coat anodizing adds 25–80 μm wear-resistant oxide to aluminum medical device housings and instrument handles with custom color coding for surgical set identification and sterile tray organization.

Mirror polishing achieves Ra 0.05–0.2 μm on titanium and cobalt-chrome implant surfaces, minimizing tissue irritation, reducing bacterial adhesion, and improving biocompatible performance on patient-contact interfaces per ISO 10993 requirements.

Vibratory finishing deburrs batch quantities of small medical CNC parts — bone screws, pins, dental abutments, and suture anchors — to Ra 0.4–0.8 μm with safe rounded edges on all body-contact surfaces.
What Materials Does XC Machining Use for Medical CNC?
XC Machining CNC machines biocompatible metals and medical-grade plastics for surgical instruments, implants, device housings, and laboratory components. Each material below includes applicable biocompatibility standards, typical medical applications, and machining characteristics. Material certifications with heat lot traceability and mill test reports are provided with every medical order.
Durable, lightweight grades (6061, 7075, 2024) used for precision medical parts and devices.
Surgical-grade stainless steel 316L (implant-contact per ASTM F138), 17-4 PH (instrument handles), and 304 (device housings) with autoclave sterilization compatibility and passivation per ASTM A967.
Mild steel 1018 and 1045 for non-patient-contact applications: medical jigs, test fixtures, manufacturing tooling, and equipment mounting brackets only.
Tool steel D2, O1, and H13 for medical injection mold inserts, stamping dies, and surgical instrument forming tools — not for patient-contact parts.
Brass C360 and C464 for medical fluid connectors, valve bodies, and low-friction instrument mechanism components requiring excellent machinability.
Copper H59 and H60 for antimicrobial medical touch surfaces, electrical contacts in diagnostic equipment, and thermal management components in imaging devices.
Biocompatible titanium Grade 2 (CP) and Grade 5 (Ti-6Al-4V per ASTM F136) for orthopedic implants, dental abutments, spinal fixation hardware, and reusable surgical instruments.

Our Medical CNC Prototype Manufacturing Capabilities
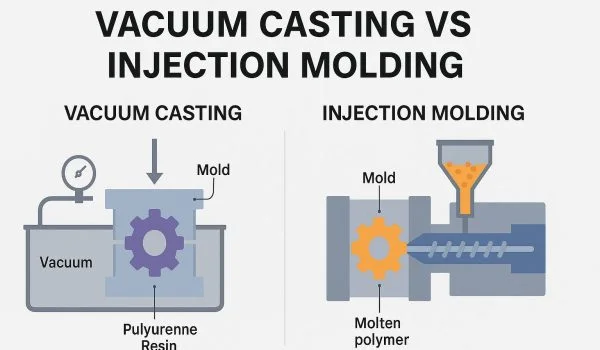
Provides injection molding for prototypes and custom parts, emphasizing expert support, competitive pricing, and faster production cycles.
Get reliable vacuum casting services for high-quality prototypes and production parts at competitive prices. XinCheng offers highly detailed casting parts with consistent quality.
Uses cutting, bending, and forming techniques to produce precision metal components suited for industrial, commercial, and manufacturing applications.
Specialist Industries
Bring Your Designs to Life with XC Machining
- Expertise Shared Widely
- Market Reach Expanded
- Advanced Technology Integrated
- Collaborative Innovation Opportunities
- All uploads are secure and confidential
Medical CNC Machining Frequently Asked Questions
When Is CNC Machining the Right Choice for Medical Device Parts?
Choose CNC machining for medical parts when: tolerances tighter than ±0.05 mm are required, the material is a biocompatible metal or engineering plastic (titanium, CoCr, 316L, PEEK, UHMWPE), the geometry is complex and cannot be injection molded, production volumes are below 10,000 units, or material certifications with full traceability are needed. Common medical CNC parts include surgical instruments, orthopedic fixation hardware, dental implants, spinal cages, and diagnostic device housings.
What Is the Medical CNC Machining Process from File to Finished Part?
The medical CNC process at XC Machining follows six steps: (1) upload STEP/IGES file, (2) DFM review for wall thickness, tool access, and material selection within 12 hours, (3) G-code programming optimized for the selected biocompatible material, (4) CNC milling or turning on calibrated machines with in-process CMM inspection, (5) post-machining finishing — passivation, electropolishing, or polishing per specification, (6) final inspection with dimensional report, material certificate with heat lot number, and certificate of conformance. Total lead time: 5–15 business days.
Can CNC Machining Produce Complex Geometries for Orthopedic and Dental Implants?
Yes. XC Machining 5-axis CNC machines produce contoured femoral condyle surfaces, anatomically shaped tibial tray profiles, porous bone-ingrowth textures, multi-angle pedicle screw channels, and undercut dental abutment geometries in titanium Grade 5 (Ti-6Al-4V), cobalt-chrome (CoCr per ASTM F75), and PEEK. Single-setup 5-axis machining eliminates re-fixturing, maintaining ±0.0127 mm accuracy across all implant contact surfaces and reducing the risk of cumulative positioning error.
What Quality Certifications and Standards Apply to Medical CNC Parts?
Medical CNC parts must comply with ISO 13485 (medical device quality management), FDA 21 CFR Part 820 (US quality system regulation), ISO 10993 (biocompatibility evaluation), and GMP (Good Manufacturing Practice). XC Machining is ISO 9001:2015 certified and produces medical components following documented quality procedures with full material traceability, heat lot tracking, CMM inspection records, and certificates of conformance per customer requirements. Contact us to discuss your specific regulatory and certification needs.